1. What is CINGAL?
CINGAL® is the first and only approved combination viscosupplement formulated to provide the benefit of a cross-linked hyaluronic acid and a fast-acting steroid.
CINGAL® is a novel combination HA plus steroid single-injection treatment for the pain associated with osteoarthritis. CINGAL® combines the high molecular weight cross-linked hyaluronic acid (HA) formulation of MONOVISC® with a corticosteroid for additional short-term pain relief.
CINGAL® is indicated as a viscoelastic supplement or a replacement for synovial fluid in human joints. CINGAL® is well suited for rapid and long term relief of the symptoms of human joint dysfunctions such as osteoarthritis.
2. The unique function of CINGAL

3. Fasting Acting. Long Lasting.
The unique combination of high concentration cross-linked hyaluronic acid formulation with a well-established FDA approved steroid provides rapid pain relief that lasts through 6 months.
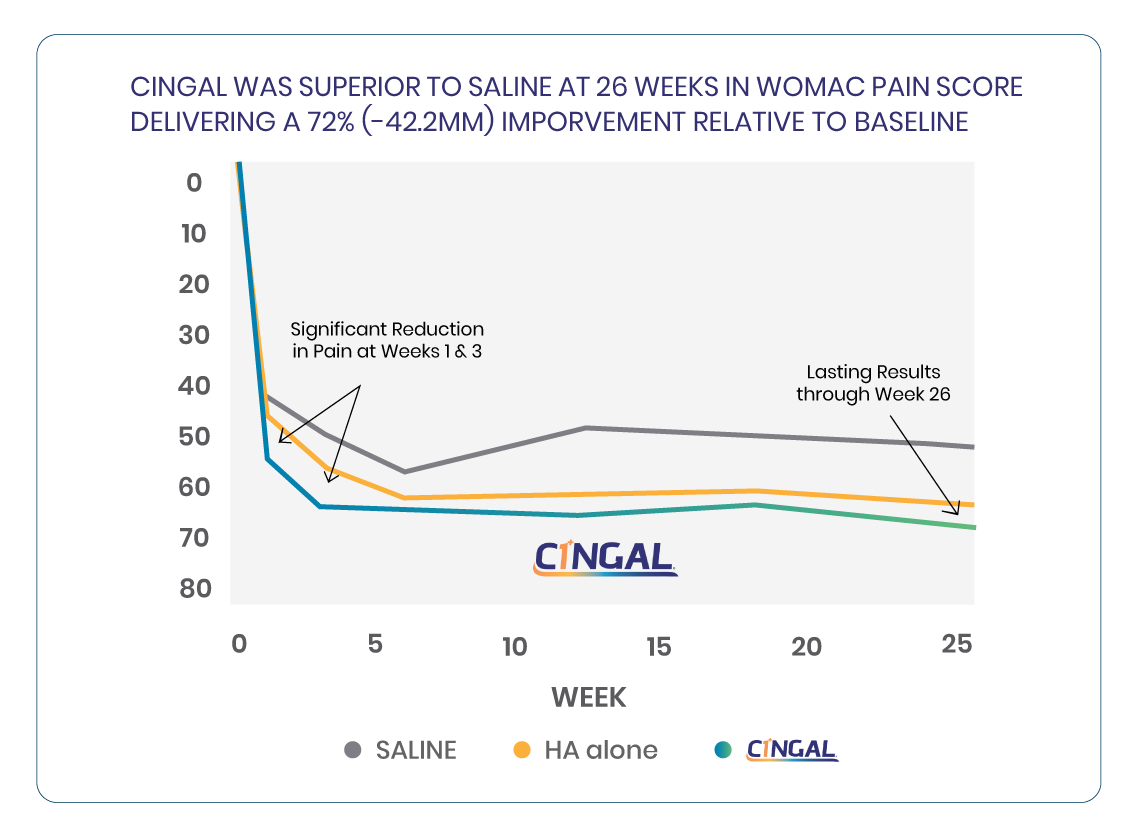
- CINGAL delivers faster pain relief compared to a leading single-injection HA viscosupplement, providing statistically significant reduction in pain at Weeks 1 and 3 relatives to HA viscosupplement and saline1
- CINGAL patients experienced long lasting pain relief, demonstrating statistically significant reduction in pain through Week 26 relative to saline1
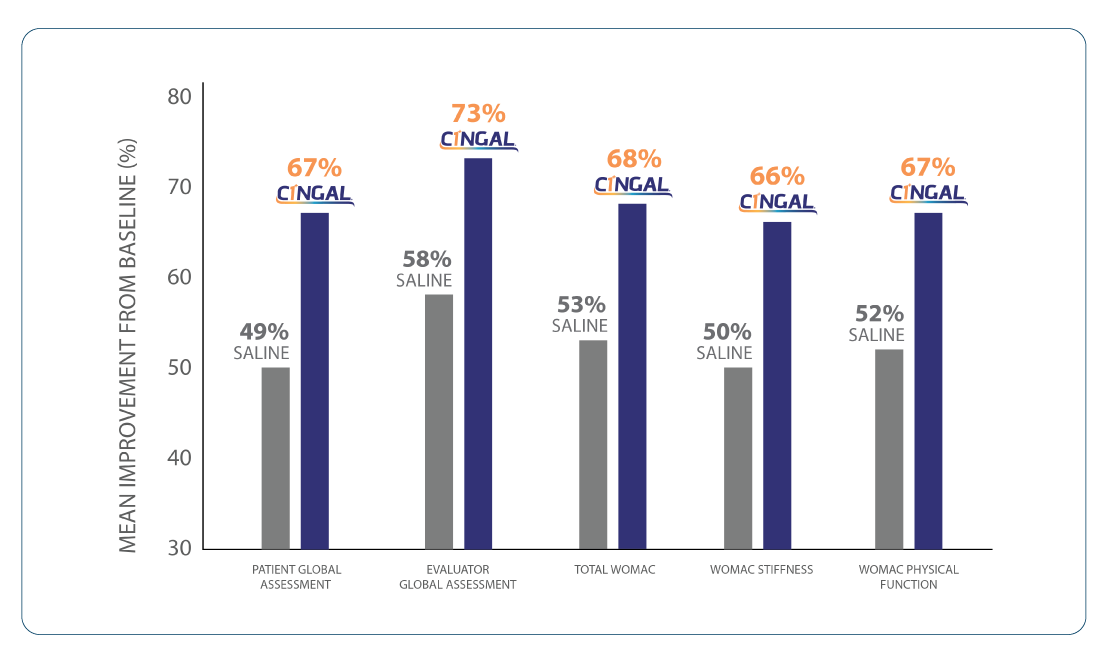
4. CINGAL shows highly statistically significant improvement compared to Saline at all secondary endpoints at 26 weeks.1
Reference 1: CINGAL 13-01, a randomized, double-blind, placebo-controlled, active comparator Phase III study. Anika Therapeutics, Inc.: study sponsor, Dr. Laszlo Hangody: global principal investigator, SynteractHCR: CRO.
5. Is retreatment clincally safe?
A retreatment was conducted in CINGAL 13-02 after the original Cingal study, CINGAL 13-01. The safety of a repeat injection of CINGAL was confirmed in the CINGAL 13-02 study.
In the CINGAL 13-02 study, 94 patients who had received CINGAL initially (from CINGAL 13-01), received an open-label injection of CINGAL 6 months after first injection. The primary end point of this study was adverse events (AEs). The key findings of the study were:
- A low number of subjects (4.3%) experienced an adverse event (AE) related to the study injection. The observed AEs were mainly associated with viscosupplements (arthralgia, injection site pain, swelling, and erythema), and over 95% were considered ‘mild’ or ‘moderate’ in severity. All AEs were transitory, resolving without treatment.
- The AE rate associated with CINGAL was found to be consistent across both first-time and repeat injection studies. There were no statistically significant differences between the AE profile of participants in the CINGAL 13-01 study (single injection) and those in the CINGAL 13-02 study (repeat injection).
Reference 2: CINGAL 13-02, an open-label, follow-on study to Cingal 13-01. Anika Therapeutics, Inc.: study sponsor, Dr. Laszlo Hangody: global principal investigator, SynteractHCR: CRO.
Cingal product information
For more information on Cingal, please download the Cingal brochure.



